 |
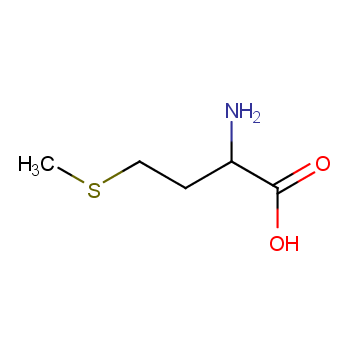
DL-Methionine
- Iupac Name:2-amino-4-methylsulfanylbutanoic acid
- CAS No.: 59-51-8
- Molecular Weight:149.21100
- Modify Date.: 2022-11-06 21:20
- Introduction: Methionine (2-amino-4(methylthio)-butanoic acid) is one of the essential amino acids, which are used by our bodies to make proteins and can only be supplied by diets. Methionine is found in metal, fish, and dairy products, and it plays an important role in many cell functions.With an asymmetric carbon, methionine molecules can be D (Dextrogyre) or L (Levogyre), optical isomers. L-methionine is the biologically active form of methionine in proteins. D-methionine is converted into L-methionine by enzymatic processes inside animal bodies in such a way that L- and D-methionine are equivalent for animal nutrition. D,L-methionine (DLM) is the most common form of commercialized methionine, composed of the racemic mixture of D and L-methionine. D,L-methionine is commercialized as a white crystalline powder, containing 99% of active substance, or in a brown liquid form with 40% purity, made from the dissolution of D,L-methionine in sodium hydroxide.Methionine is used to prevent liver damage in acetaminophen (Tylenol) poisoning. It is also used to increase the acidity of urine, to treat liver disorders, and to improve wound healing. Furthermore, it is used for the treatment of depression, alcoholism, allergies, asthma, copper poisoning, radiation side effects, schizophrenia, drug withdrawal, and Parkinson's disease. Methionine is registered as an ingredient for use in cosmetics as antistatic and for skin conditioning (Commission Decision 2006/257/EC).D,L-methionine is used in animal feeds for poultry and pigs, as well as in diets for cats and dogs. D,L-methionine technically pure protected with copolymer vinylpyridine/styrene, and D,L-methionine protected with ethylcellulose when used as a feed additive for ruminants, are currently authorized for use for all animal species and ruminants, respectively, by Regulation (EU) No 469/2013.
View more+
1. Names and Identifiers
- 1.1 Name
- DL-Methionine
- 1.2 Synonyms
(R,S)-2-Amino-4-methylsulfanyl-butyricacid (RS)-2-Amino-3-methylthiobutansαure Amurex Cynaron DL-2-Amino-4-(methylthio)butanoic acid Dyprin EINECS 200-432-1 L-MET Meonine MET Metion Metione MFCD00063097 Neston Pedameth Petameth Thiomedon Urimeth
- View all
- 1.3 CAS No.
- 59-51-8
- 1.4 CID
- 876
- 1.5 EINECS(EC#)
- 200-432-1
- 1.6 Molecular Formula
- C5H11NO2S (isomer)
- 1.7 Inchi
- InChI=1S/C5H11NO2S/c1-9-3-2-4(6)5(7)8/h4H,2-3,6H2,1H3,(H,7,8)
- 1.8 InChIkey
- FFEARJCKVFRZRR-UHFFFAOYSA-N
- 1.9 Canonical Smiles
- CSCCC(C(=O)O)N
- 1.10 Isomers Smiles
- CSCCC(C(=O)O)N
2. Properties
- 2.1 Density
- 1.34
- 2.1 Melting point
- 270-273oC
- 2.1 Boiling point
- 306.9oC at 760 mmHg
- 2.1 Refractive index
- 1.5216 (estimate)
- 2.1 Flash Point
- 139.4oC
- 2.1 Precise Quality
- 149.05100
- 2.1 PSA
- 88.62000
- 2.1 logP
- 0.85170
- 2.1 Solubility
- 2.9 g/100 mL (20 oC)
- 2.2 Appearance
- white crystalline powder with a faint odor
- 2.3 Storage
- Ambient temperatures.
- 2.4 Chemical Properties
- White crystalline powder
- 2.5 Color/Form
- Minute hexagonal plates from dilute alcohol
Colorless or white, lustrous plates or as white, crystalline powder
- 2.6 Physical
- Solid
- 2.7 pKa
- 2.13(at 25℃)
- 2.8 Water Solubility
- 1 M HCl: 0.5?M at?20?°C, clear, colorless | 2.9 g/100 mL (20 oC)
- 2.9 Spectral Properties
- MAX ABSORPTION (0.01 N HCL): 208 NM (SHOULDER) (LOG E= 3.2)
Specific optical rotation: 22.5 deg at 25 deg C/D (1 N HCl)
Specific optical rotation: -8.2 deg at 25 deg C/D (c = 0.8); +23.40 at 20 deg C/D (c = 5.0 in 3.0N HCl)
MASS: 54346 (NIST/EPA/MSDC Mass Spectral Database, 1990 version)
- 2.10 Stability
- Stable. Incompatible with strong oxidising agents.
- 2.11 StorageTemp
- 2-8°C
3. Use and Manufacturing
- 3.1 Definition
- ChEBI: A sulfur-containing amino acid that is butyric acid bearing an amino substituent at position 2 and a methylthio substituent at position 4.
- 3.2 GHS Classification
- Signal: Warning
GHS Hazard Statements
The GHS information provided by 1 company from 1 notification to the ECHA C&L Inventory.
H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation]
H319 (100%): Causes serious eye irritation [Warning Serious eye damage/eye irritation]
Precautionary Statement Codes
P264, P280, P302+P352, P305+P351+P338, P321, P332+P313, P337+P313, and P362
- 3.3 Purification Methods
- Crystallise it from hot water or EtOH. Also purify it by dissolving it in H2O and passing through an Amberlite IR-120 resin (NH4+ form). The eluate is concentrated and then passed through Amberlite IR-4B resin, and this eluate is evaporated to dryness. The residue is washed with EtOH, then Me2CO, dried and recrystallised from aqueous EtOH (colourless plates) [Baddiley & Jamieson J Chem Soc 4283 1954]. [Greenstein & Winitz The Chemistry of the Amino Acids J. Wiley, Vol 3 p 2125 1961, Beilstein 4 IV 3190.]
- 3.4 Usage
- An essential nonpolar amino acid with oxidative stress defense properties
4. Safety and Handling
- 4.1 Hazard Codes
- Xi
- 4.1 Risk Statements
- R33
- 4.1 Safety Statements
- S24/25
- 4.1 Exposure Standards and Regulations
- L-Methionine is a food additive permitted for direct addition to food for human consumption, as long as 1) the quantity of the substance added to food does not exceed the amount reasonably required to accomplish its intended physical, nutritive, or other technical effect in food, and 2) any substance intended for use in or on food is of appropriate food grade and is prepared and handled as a food ingredient.
Drug products containing certain active ingredients offered over-the-counter (OTC) for certain uses. A number of active ingredients have been present in OTC drug products for various uses, as described below. However, based on evidence currently available, there are inadequate data to establish general recognition of the safety and effectiveness of these ingredients for the specified uses: methionine is included in weight control drug products.
Methionine used as a nutrient and/or dietary supplement in animal drugs, feeds, and related products is generally recognized as safe when used in accordance with good manufacturing or feeding practice. - View all
- 4.2 Octanol/Water Partition Coefficient
- log Kow = -1.87
- 4.3 DisposalMethods
- SRP: Expired or waste pharmaceuticals shall carefully take into consideration applicable DEA, EPA, and FDA regulations. It is not appropriate to dispose by flushing the pharmaceutical down the toilet or discarding to trash. If possible return the pharmaceutical to the manufacturer for proper disposal being careful to properly label and securely package the material. Alternatively, the waste pharmaceutical shall be labeled, securely packaged and transported by a state licensed medical waste contractor to dispose by burial in a licensed hazardous or toxic waste landfill or incinerator.
SRP: At the time of review, regulatory criteria for small quantity disposal are subject to significant revision, however, household quantities of waste pharmaceuticals may be managed as follows: Mix with wet cat litter or coffee grounds, double bag in plastic, discard in trash.
SRP: Criteria for land treatment or burial (sanitary landfill) disposal practices are subject to significant revision. Prior to implementing land disposal of waste residue (including waste sludge), consult with environmental regulatory agencies for guidance on acceptable disposal practices. - View all
- 4.4 RIDADR
- NONH for all modes of transport
- 4.4 Safety Profile
- Moderately toxic by ingestion and other routes. An experimental teratogen. Experimental reproductive effects. When heated to decomposition it emits toxic fumes of SOx and NOx. See also 1-METHIONINE.
- 4.5 Formulations/Preparations
- NF, feed 98%
USP and FCC grades; 99% feed grade
Racemate mixture of D- and L- methionine
- 4.6 WGK Germany
- 2
- 4.6 RTECS
- PD0457000
- 4.6 Safety
-
Moderately toxic by ingestion and other routes. An experimental teratogen. Experimental reproductive effects. When heated to decomposition it emits toxic fumes of SOx and NOx.?
Hazard Codes:?Xi
Risk Statements: 33-36/37/38?
R33:Danger of cumulative effects.?
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 24/25-36-26?
S24/25:Avoid contact with skin and eyes.?
S36:Wear suitable protective clothing.?
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
WGK Germany: 2
RTECS: PD0457000
F: 10-23
- 4.7 Specification
-
?Racemethionine , its cas register number is 59-51-8. It also can be called?(+-)-Methionine ; Acimetion ; Banthionine ; Cynaron ; DL-2-Amino-4-(methylthio)-butyric acid ; DL-Methioninum ; Dyprin ; Lobamine ; alpha-Amino-gamma-(methylthio)butyric acid ; alpha-Amino-gamma-methylmercaptobutyric acid ; alpha-Amino-gamma-methylmercaptobutyric acid (VAN)?.It is a?white crystalline powder with a faint odor.
- 4.8 Toxicity
-
| Organism |
Test Type |
Route |
Reported Dose (Normalized Dose) |
Effect |
Source |
| mouse |
LDLo |
intraperitoneal |
1500mg/kg (1500mg/kg) |
? |
Yakuri to Chiryo. Pharmacology and Therapeutics. Vol. 5, Pg. 2041, 1977. |
| mouse |
LDLo |
intravenous |
300mg/kg (300mg/kg) |
? |
Yakuri to Chiryo. Pharmacology and Therapeutics. Vol. 5, Pg. 2041, 1977. |
| mouse |
LDLo |
oral |
4gm/kg (4000mg/kg) |
? |
Yakuri to Chiryo. Pharmacology and Therapeutics. Vol. 5, Pg. 2041, 1977. |
| rat |
LD |
intravenous |
> 400mg/kg (400mg/kg) |
? |
Yakuri to Chiryo. Pharmacology and Therapeutics. Vol. 5, Pg. 2041, 1977. |
| rat |
LD |
oral |
> 5gm/kg (5000mg/kg) |
? |
Yakuri to Chiryo. Pharmacology and Therapeutics. Vol. 5, Pg. 2041, 1977. |
| rat |
LDLo |
intraperitoneal |
2gm/kg (2000mg/kg) |
? |
Yakuri to Chiryo. Pharmacology and Therapeutics. Vol. 5, Pg. 2041, 1977. |
- View all
5. MSDS
2.Hazard identification
2.1 Classification of the substance or mixture
Not classified.
2.2 GHS label elements, including precautionary statements
| Pictogram(s) | No symbol. |
| Signal word | No signal word. |
| Hazard statement(s) | none |
| Precautionary statement(s) | |
| Prevention | none |
| Response | none |
| Storage | none |
| Disposal | none |
2.3 Other hazards which do not result in classification
none
7. Synthesis Route
59-51-8Total: 40 Synthesis Route
8. Other Information
- 8.0 Usage
- Production of volatile compounds related to the flavour of foods from the Strecker degradation of DL-methionine. Addition of methionine in appropriate amounts to these foods might be expected to improve protein value. Economically, DL-methionine would be preferable.
- 8.1 Merck
- 14,5975
- 8.2 BRN
- 636185
- 8.3 Description
- Methionine (2-amino-4(methylthio)-butanoic acid) is one of the essential amino acids, which are used by our bodies to make proteins and can only be supplied by diets. Methionine is found in metal, fish, and dairy products, and it plays an important role in many cell functions.
With an asymmetric carbon, methionine molecules can be D (Dextrogyre) or L (Levogyre), optical isomers. L-methionine is the biologically active form of methionine in proteins. D-methionine is converted into L-methionine by enzymatic processes inside animal bodies in such a way that L- and D-methionine are equivalent for animal nutrition. D,L-methionine (DLM) is the most common form of commercialized methionine, composed of the racemic mixture of D and L-methionine. D,L-methionine is commercialized as a white crystalline powder, containing 99% of active substance, or in a brown liquid form with 40% purity, made from the dissolution of D,L-methionine in sodium hydroxide.
Methionine is used to prevent liver damage in acetaminophen (Tylenol) poisoning. It is also used to increase the acidity of urine, to treat liver disorders, and to improve wound healing. Furthermore, it is used for the treatment of depression, alcoholism, allergies, asthma, copper poisoning, radiation side effects, schizophrenia, drug withdrawal, and Parkinson's disease. Methionine is registered as an ingredient for use in cosmetics as antistatic and for skin conditioning (Commission Decision 2006/257/EC).
D,L-methionine is used in animal feeds for poultry and pigs, as well as in diets for cats and dogs. D,L-methionine technically pure protected with copolymer vinylpyridine/styrene, and D,L-methionine protected with ethylcellulose when used as a feed additive for ruminants, are currently authorized for use for all animal species and ruminants, respectively, by Regulation (EU) No 469/2013. - View all
- 8.4 References
- [1] DL-Methionine from Methional and Hyddrogen Cyanide – Cost Analysis – Methionine E11A (2016)
[2] http://www.rxlist.com/methionine/supplements.htm
[3] http://onlinelibrary.wiley.com/doi/10.2903/j.efsa.2012.2623/abstract
- 8.5 Description
- Methionine is an α-amino acid with the chemical formula HO2CCH(NH2)CH2CH2SCH3. This essential amino acid is classified as nonpolar. This amino-acid is coded by the initiation codon AUG which indicates mRNA's coding region where translation into protein begins.
- 8.6 Chemical Properties
- White crystalline powder
- 8.7 Chemical Properties
- White, crystalline platelets or powder having a characteristic odor. One g dissolves in about 30 mL of water. It is soluble in dilute acids and in solutions of alkali hydroxides. It is very slightly soluble in alcohol, and practically insoluble in ether. It is optically inactive. The pH of a 1 in 100 solution is between 5.6 and 6.1. This substance may be prepared by addition of methanethiol to acrolein; by chemical conversion of methylthiopropionic aldehyde.
- 8.8 Chemical Properties
- d,l-Methionine has a characteristic odor. It is an essential amino acid and is also used as a nutrient and dietary supplement.
- 8.9 Occurrence
- High levels of methionine can be found in eggs, sesame seeds, Brazil nuts, fish, meats and some other plant seeds; methionine is also found in cereal grains. Most fruits and vegetables contain very little of it. Most legumes are also low in methionine. Racemic methionine is sometimes added as an ingredient to pet foods.
- 8.10 Uses
- An essential nonpolar amino acid with oxidative stress defense properties
- 8.11 Uses
- DL-Methionine is sometimes given as a supplement to dogs; it helps keep dogs from damaging grass by reducing the pH of the urine.
Methionine is allowed as a supplement to organic poultry feed under the US certified organic program.
- 8.12 Definition
- ChEBI: A sulfur-containing amino acid that is butyric acid bearing an amino substituent at position 2 and a methylthio substituent at position 4.
- 8.13 Preparation
- By addition of methanethiol to acrolein; by chemical conversion of methylthiopropionic aldehyde.
- 8.14 Brand name
- Pedameth (Forest).
- 8.15 Biosynthesis
- As an essential amino acid, methionine is not synthesized de novo in humans, who must ingest methionine or methioninecontaining proteins. In plants and microorganisms, methionine is synthesized via a pathway that uses both aspartic acid and cysteine. First, aspartic acid is converted via β-aspartyl-semialdehyde into homoserine, introducing the pair of contiguous methylene groups. Homoserine converts to O-succinyl homoserine, which then reacts with cysteine to produce cystathionine, which is cleaved to yield homocysteine. Subsequent methylation of the thiol group by folates affords methionine. Both cystathionine-γ-synthase and cystathionine- β-lyase require pyridoxyl-5′-phosphate as a cofactor, whereas homocysteine methyltransferase requires vitamin B12 as a cofactor.
- 8.16 Biotechnological Production
- DL-Methionine is the second amino acid that is almost exclusively manufactured by chemical synthesis. The process used today was originally developed by Werner Schwarze at Degussa in the 1940s, and has been continually improved and refined since. Today DL-methionine is manufactured in several plants, each with a capacity of more than 100,000 tonnes. To operate the process on an industrial scale also requires back-integration into the key hazardous raw materials acrolein, methyl mercaptan, and hydrogen cyanide. After the formation of the hydantoin, the key step is alkaline hydrolysis of the hydantoin, to produce methionine directly in up to 95 % yield based on acrolein.
- 8.17 Biological Functions
- Together with cysteine, methionine is one of two sulfurcontaining proteinogenic amino acids. Its derivative S-adenosyl methionine (SAM) serves as a methyl donor. Methionine is an intermediate in the biosynthesis of cysteine, carnitine, taurine, lecithin, phosphatidylcholine, and other phospholipids. Improper conversion of methionine can lead to atherosclerosis.
This amino acid is also used by plants for synthesis of ethylene. The process is known as the Yang Cycle or the methionine cycle.
Methionine is one of only two amino acids encoded by a single codon (AUG) in the standard genetic code (tryptophan, encoded by UGG, is the other). The codon AUG is also the most common eukaryote "Start" message for a ribosome that signals the initiation of protein translation from mRNA when the AUG codon is in a Kozak consensus sequence. As a consequence, methionine is often incorporated into the N-terminal position of proteins in eukaryotes and archaea during translation, although it can be removed by post-translational modification. In bacteria, the derivative Nformylmethionine is used as the initial amino acid. - View all
- 8.18 General Description
-
DL-Methionine is an essential amino acid containing sulphur. Methionine consists of an asymmetric carbon and exists as D (dextrogyre) and L (levogyre) optical isomers. The L-methionine is considered as biologically active. The racemic mixture of D and L-isomers forms DL-methionine, which is the commercially available methionine.
9. Computational chemical data
- Molecular Weight: 149.21100g/mol
- Molecular Formula: C5H11NO2S
- Compound Is Canonicalized: True
- XLogP3-AA: null
- Exact Mass: 149.05104977
- Monoisotopic Mass: 149.05104977
- Complexity: 97
- Rotatable Bond Count: 4
- Hydrogen Bond Donor Count: 2
- Hydrogen Bond Acceptor Count: 4
- Topological Polar Surface Area: 88.6
- Heavy Atom Count: 9
- Defined Atom Stereocenter Count: 0
- Undefined Atom Stereocenter Count: 1
- Defined Bond Stereocenter Count: 0
- Undefined Bond Stereocenter Count: 0
- Isotope Atom Count: 0
- Covalently-Bonded Unit Count: 1
- CACTVS Substructure Key Fingerprint: AAADccBiMABAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAHgQQCAAACCjFwASCCABAAggIAACQCAAAAAAAABAAAIGAAAACABAgAAAAQAAEEAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAA==
10. Question & Answer
-
Methionine, an essential amino acid, is found in meat, fish, and dairy products. It plays a crucial role in building molecules necessary for cell and DNA function. Exploring the Health Benefits Amino..
-
DL-Methionine, also known as methionine, is an important amino acid that can improve the nutritional value of feed and help animals grow quickly in a short period of time. It is an essential amino aci..
-
DL-Methionine, first isolated and purified by American biochemist John Howard Mueller in 1921, is one of the two sulfur-containing amino acids. It is the only sulfur-containing essential amino acid fo..
-
DL-Methionine, abbreviated as Met, is one of the basic units for protein synthesis and the only sulfur-containing amino acid and limiting amino acid. Animals can significantly promote growth and short..
11. Recommended Suppliers
-
- Products:Chemical products
- Tel:86-571-88938639
- Email:sales-gc@dycnchem.com
-
- Products:Our company specializes in the research and development of fine chemicals, food additives and pharmaceutical raw materials
- Tel:86-13126149-050
- Email:linda@hbsaisier.cn
-
- Products:chemical
- Tel:86-44202508-1565566465131
- Email:13210232125@qd-dm.com
-
- Products:Plastic particles, chemical products
- Tel:+86-22-15233218311
- Email:emily@furuntongda.com
-
- Products:API',s intermediates,fine chemicals and plant extracts.
- Tel:86-917-3909592
- Email:gksales1@gk-bio.com
12. Realated Product Infomation





